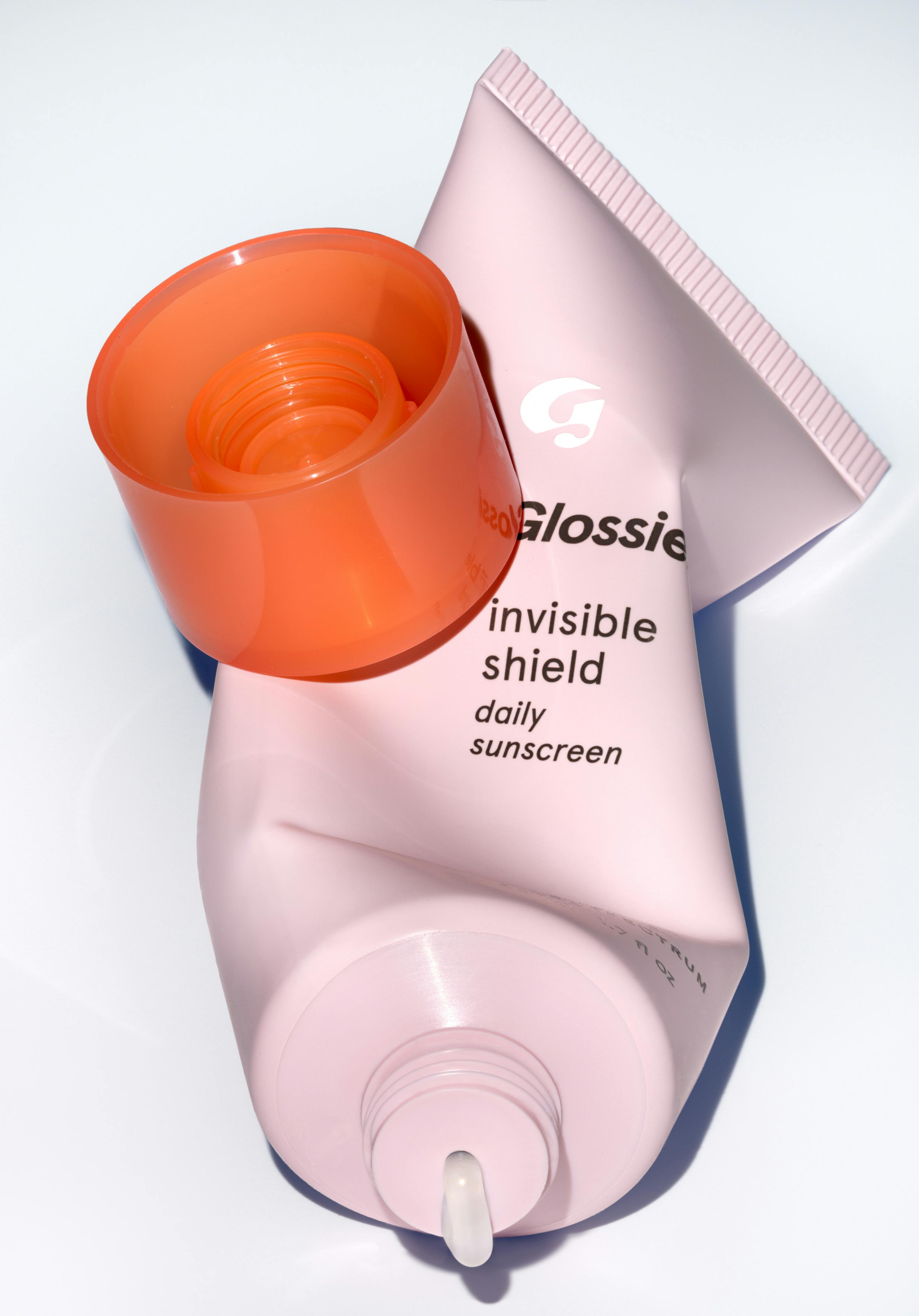
Score free shipping on qualifying orders! For more information on shipping rates and timelines, please visit our international page
The Balm Dotcom Trio: for a limited time, choose 3 flavors of our universal salve for $30.
Free standard shipping on orders $65 CAD+. For more information on shipping rates and timelines, please visit our international page
Livraison gratuite $65 CAD+. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders $110 AUD or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders R$670 or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders ¥990 or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 480kr or more. For more information on shipping rates and timelines, please visit our international page
Livraison gratuite 65€ +. Score free shipping on qualifying orders! For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders ₹11,230 or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders ₪515 or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders ₩180,035 or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders MX$1,320. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders 65€ or more. For more information on shipping rates and timelines, please visit our international page
Free express shipping on orders 900kr or more. For more information on shipping rates and timelines, please visit our international page
Free standard shipping on orders $120 NZD or more. For more information on shipping rates and timelines, please visit our international page